Complexes of environmentally benign ligands and cations
|
|
Amino acids and related compounds are good nucleophiles in their neutral or deprotonated form. Their chemical variety with respect to binding modes and their stereochemistry make them interesting ligands. Their complexes with biocompatible cations are promising targets for basic research which may even be studied in high school labs. Such compounds may find applications as functional materials of low toxicity or as food additives.
|
|
My youngest co-author ever contributed to this article during her time in high school: The ephemeral dihydrate of sulfanilic acid.
S. Langenstück, C. Zhao, U. Englert; Acta Crystallogr. 2018, C74, 7-12.
Silver Complexation by Metallacryptates.
K. Lamberts, M. Tegoni, X. Jiang, H. Kou, U. Englert; Dalton Trans. 2016, 45, 284-295.
Incidental Polymorphism, Non-Isomorphic and Isomorphic Substitution in Calcium-Valine Coordination Polymers.
K. Lamberts, U. Englert; Crystals 2015, 5, 261-272.
Biguanide and squaric acid as pH-dependent building blocks in crystal engineering.
M.-D. Serb, I. Kalf, U. Englert; CrystEngComm 2014, 16, 10631-10639.
An unusual linker and an unexpected node: CaCl2 dumbbells linked by proline to form square lattice networks.
K. Lamberts, S. Porsche, B. Hentschel, T. Kuhlen, U. Englert; CrystEngComm 2014, 16, 3305-3311.
N-(6-Methyl-2-pyridyl)mesitylenesulfonamide: An Efficient Template for Polyiodides.
F. Pan, U. Englert; Cryst. Growth Des. 2014, 14, 1057-1066.
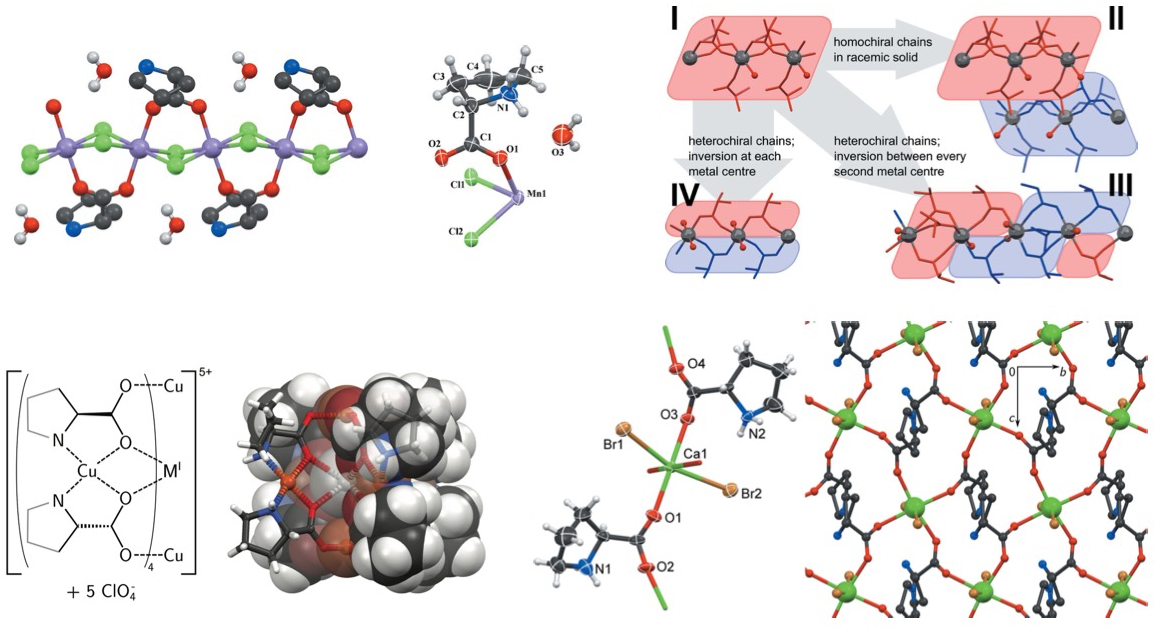
Structures from MnX2 and proline: isomorphous racemic compounds and a
series of chiral non-isomorphous chain polymers.
K. Lamberts, U. Englert; Acta Crystallogr. 2012, B68, 610-618.
Disclaimer to Web Site Contents
Ulli(2020-01-02, 2020-01-02)
